Pathway Charting Practice
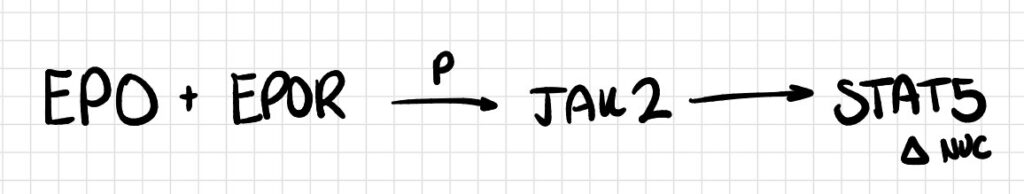
Pathway 1
Erythropoietin (EPO) binds to its homodimeric receptor (EPOR) on erythroid progenitor cells, triggering transphosphorylation of the associated JAK2 kinases. Phosphorylated JAK2 subsequently activates STAT5, which dimerizes and translocates to the nucleus to drive transcription of anti-apoptotic genes such as bcl-xL.
Chart
Pathway 2
Upon activation by double-stranded RNA, protein kinase R (PKR) phosphorylates the alpha subunit of eukaryotic initiation factor 2 (eIF2α) at Ser51. Phosphorylated eIF2α sequesters the guanine nucleotide exchange factor eIF2B, thereby globally suppressing cap-dependent translation initiation, halting viral protein synthesis.
Chart
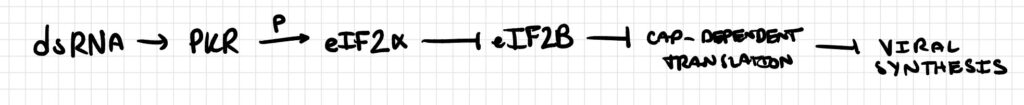
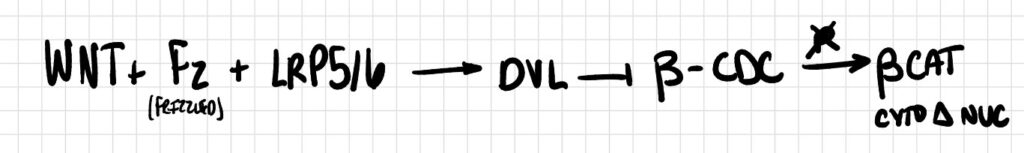
Pathway 3
Binding of Wnt ligand to its co-receptors Frizzled and LRP5/6 recruits the scaffold protein Dishevelled (DVL), inhibits the beta-catenin destruction complex (β-CDC). The β-CDC is a multimeric protein composed of APC, Axin, GSK-3β, and CK1 that normally phosphorylates and subsequent degrades β-catenin, preventing its accumulate in the cytoplasm and subsequent translocation to the nucleus.
Chart
Pathway 4
Angiotensin II (AII) binds to its AT1 receptor, a Gq-coupled GPCR, activating phospholipase C (PLC). PLC cleaves PIP2 into IP3 and DAG. The newly produced IP3 triggers calcium release from the endoplasmic reticulum, while DAG activates protein kinase C (PKC), collectively promoting vasoconstriction and aldosterone secretion.
Chart
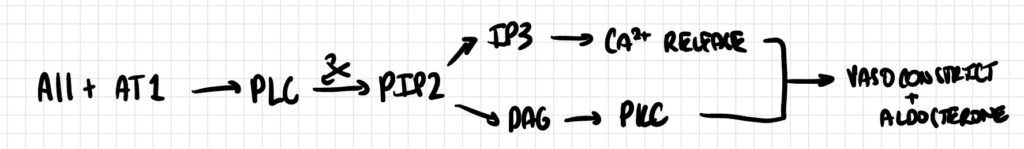
Pathway 5
Activation of the Toll-like receptor 4 (TLR4) by lipopolysaccharide (LPS) on the surface of macrophages initiates a MyD88-dependent signaling cascade. MyD88 recruits IRAK4, which phosphorylates and activates IRAK1. IRAK1 then associates with TRAF6, an E3 ubiquitin ligase that generates K63-linked polyubiquitin chains. These chains serve as scaffolds that recruit and activate the TAK1 kinase complex, which phosphorylates the IKK complex. IKK phosphorylates IκBα, targeting it for proteasomal degradation and thereby releasing NF-κB to translocate to the nucleus and induce pro-inflammatory gene expression.
Chart
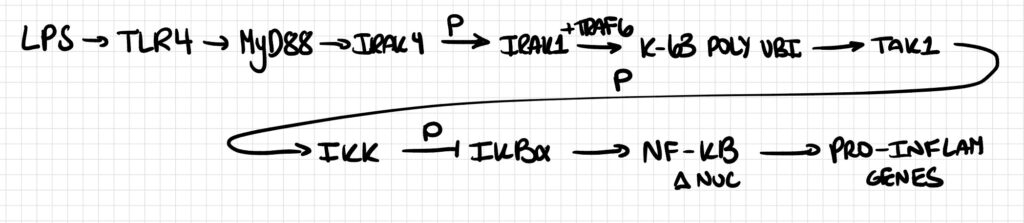
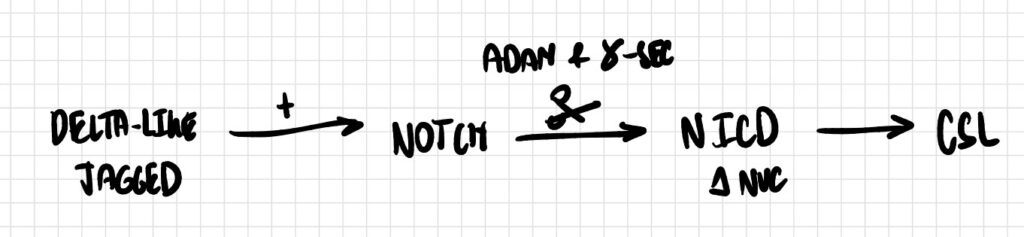
Pathway 6
The Notch receptor is activated by binding to membrane-bound ligands Delta-like or Jagged on adjacent cells. This interaction promotes successive cleavage of Notch by ADAM metalloprotease and then γ-secretase, releasing the Notch intracellular domain (NICD), which translocates to the nucleus and converts the transcriptional repressor CSL into a transcriptional activator.
Chart