Practice: Basic Passage Reading
RLC in Heart Failure
Despite recent treatment advances, heart failure is one of the leading causes of death and hospitalization in cardiovascular patients. While most treatments are focused on the role of troponin in myocyte dysfunction, newer treatments are aimed at targeting cardiac myosin regulatory light chain (RLC), a thick-filament-associated protein.
Phosphorylation of RLC was recently demonstrated to increase force development as well as Ca2+ sensitivity of skinned cardiac fibers and altered RLC phosphorylation is known to contribute to compensatory responses and contractile dysfunction in heart failure.
However, the exact mechanisms by which RLC contributes to cardiac dysfunction remain unknown hindering development of new drug treatments. To investigate this further scientists focused on the role of an exchange protein activated by cAMP (Epac) in RLC phosphorylation. Epac has two isoforms (Epac1 and Epac2) and only Epac1 is ubiquitously expressed in the heart.
To determine the contribution of Epac1 to RLC phosphorylation two mouse lines were generated: a non-transgenic control mouse (NTG) and a transgenic mouse that overexpresses Epac1 (Epac1TG). First the specificity of Epac1 was tested by incubating cardiac tissue from NTG mice with 8-CPT-AM, a known Epac1 activator or 8-CPT-AM and H-89, a highly specific inhibitor of protein kinase A (PKA).
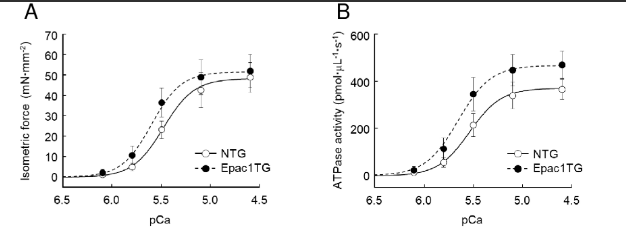
Figure 1: Relative phosphorylation level of different proteins (RLC, troponin I (TnI), troponin T (TnT), and myosin binding protein C (MyBP-C)) in the presence of buffer only (CRTL), presence of 8CPT-AM, and presence of 8CPT-AM and H-89.
Following the tests for specificity the force generation and myofibrillar ATPase activity was accessed at varied calcium concentrations expressed as pCa (–log[Ca²⁺]) in NTG and Epac1TG mice.
Adapted from: Ohnuki Y, Suita K, Ishikawa M, Mototani Y, Nariyama M, Ito A, et al. (2025) Epac1 increases myosin regulatory light-chain phosphorylation, energetic cost of contraction, and susceptibility to heart failure. PLoS One
